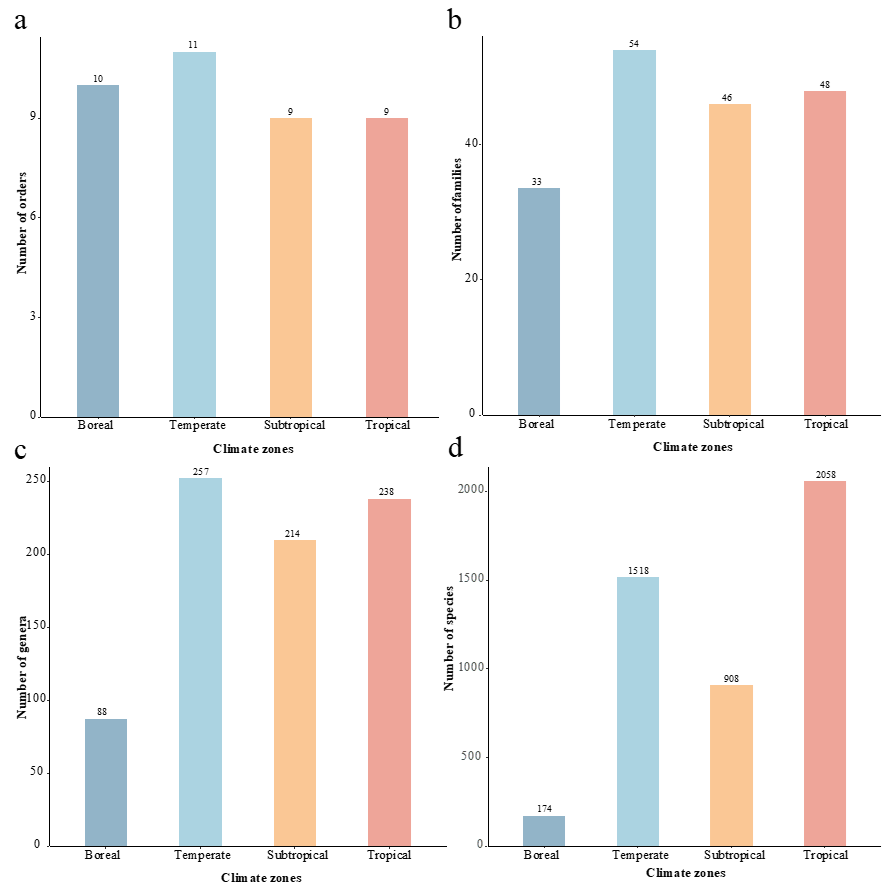-
Fungi are among the most diverse groups of organisms on Earth, playing a crucial role in ecosystem processes and func-tioning (Han et al. 2023; Wang et al. 2023a; Zhao et al. 2023a; Abrego et al. 2024; Dong et al. 2024, 2025; He et al. 2024a; Hyde et al. 2024a; Bao et al. 2025; Hibbett et al. 2025; Hongsanan et al. 2025; Lin et al. 2025; Samaradiwakara et al. 2025), and their economic values to society are estimated at 54.57 trillion USD (Niego et al. 2023). Various studies have explored the earth’s fungal species diversity, and a conserva-tive estimate suggested that the number of fungal species ranged from 2.2 to 3.8 million, and revised estimate of 2 to 3 million fungal species with a “best estimate” at 2.5 million (Hawksworth & Lücking 2017; Baldrian et al. 2022; Bhunjun et al. 2022; Niskanen et al. 2023; Hyde et al. 2024a), while high-throughput sequencing indicated 11.7 to 13.2 million species (Wu et al. 2019a; Baldrian et al. 2022). However, only 202,799 fungal species have been recorded in the Fungal Names database to date (Wang et al. 2023b, https://nmdc.cn/fungalnames, accessed on 8 November 2025), accounting for 5.34–9.22 % of the conservative estimate. In recent times, with approximately 1,000–3,000 new fungal species being described annually, it suggests that many spe-cies are yet to be discovered (Bhunjun et al. 2022; Dis-sanayake et al. 2025; Hibbett et al. 2025).
Among these diverse groups, polypores are one of the most important wood-inhabiting fungi with complex morphology and ecology (Fig. 1), playing an important role in forest regenera-tion and energy cycling. The majority of polypores are sapro-trophs, breaking down key components of plant cell walls, such as lignin, cellulose, hemicellulose, and pectin by various car-bohydrate-active enzymes (Berglund et al. 2011; Krah et al. 2018; Wu et al. 2022a, 2022b). Some polypore species, such as Heterobasidion spp. and Phaeolus spp., are pathogenic or facultative parasites of living trees, causing tree mortality and consequent significant economic losses for commercial for-estry (Yuan et al. 2021, 2023; Wu et al. 2022b; Garcia et al. 2024; Cui et al. 2025a). A few polypores, such as Albatrellus spp., are known to form ectomycorrhizal relationships (Teder-soo et al. 2007; Zhou et al. 2024a). Notably, certain polypore species, such as Sanghuangporus spp. and Ganoderma spp., are used in traditional Chinese medicine, and modern studies have shown that they have anticancer, anti-inflammatory, antioxidant, cytotoxic, antiallergic, hepatoprotective, hypoten-sive, antiviral, and neuroprotective properties (Wang et al. 2022, 2023c; Blundell et al. 2023; Sułkowska-Ziaja et al. 2023; Ghobad-Nejhad et al. 2024) and are a potential source of pharmaceutical products. Additionally, a few polypore species, like Albatrellus spp. and Grifola albicans f. huishuhua X.J. Xie, are edible mushrooms (Wu et al. 2019b; Xie et al. 2024; Zhou et al. 2024a). Linnaeus (1753) introduced the binomial nomenclature for naming organisms and described several polypores, such as Coltricia perennis (L.) Murrill, Daedalea quercina (L.) Pers., Lenzites betulinus (L.) F., and Phellinus igniarius (L.) Quél. (Wang et al. 2023b, https://nmdc.cn/fungalnames/, accessed on 17 April 2024). Subsequently, the species descriptions of polypores became more systematic, with morphological traits consistently remaining a key factor in the taxonomy of these fungi. The species diversity of polypores has attracted wide-spread attention, particularly since the early 21st century, when molecular biology was widely applied in fungal taxonomy. In recent decades, the number of recognized polypore species has increased significantly through the efforts of numerous mycologists. In China, many studies have focused on pol-ypores, contributing to the description of new taxa, understanding distribution patterns, reconstructing phylogenetic relationships, estimating diver-gence times, and exploring historical biogeography (Dai 2010, 2012; Cui et al. 2019, 2024; Wu et al. 2021, 2022a, 2022b; Zhao et al. 2022, 2023b, 2024; Liu et al. 2023a, 2023b; Wang et al. 2023a, 2024a, 2024b; Zhou et al. 2023; Xu et al. 2025). Ryvarden and his colleagues have described numerous pol-ypore species globally and published a series of influential monographs, such as the North American polypores, poroid fungi of Europe, and poroid fungi of Africa (Ryvarden & Jo-hansen 1980; Ryvarden & Gilbertson 1986, 1987; Ryvarden 1991, 2000, 2004, 2015, 2016, 2018a, 2018b, 2020, 2023, 2024a, 2024b; Ryvarden & Melo 2014, 2017; Ryvarden et al. 2022; Spirin & Ryvarden 2024). In addition, the biodiversity and distribution patterns of polypores from North America and South America have been studied, with a large number of new taxa proposed, and new distributions of existing taxa (Ryvarden & Gilbertson 1986, 1987; Gilbertson 1991; Gibertoni et al. 2004, 2016; Gibertoni & Ryvarden 2014; Gilbertson & Hemmes 2004; Oliveira & Gib-ertoni 2023; Oliveira et al. 2023, 2025).
 Fig 1. Basidiomata of represented taxa of polypores. (a) Albatrellus piceiphilus. (b) Anomoloma submyceliosum. (c) Aporpium macroporum. (d) Bo-letopsis macrocarpa. (e) Bondarcevomyces taxi. (f) Coltricia perennis. (g) Favolaschia longistipitata. (h) Fistulina hepatica. (i) Fomes fomentarius. (j) Ganoderma sinense. (k) Gloeophyllum sepiarium. (l) Heterobasidion insulare. (m) Hexagonia apiaria. (n) Laetiporus xinjiangensis. (o) Lentinus arcularius. (p) Megasporoporia bannaensis. (q) Phellinus nigricans. (r) Polyporus umbellatus. (s) Porodaedalea himalayensis. (t) Radulotubus resupinatus. (u) Rhodofomes roseus. (v) Sanghuangporus sanghuang. (w) Sistotrema muscicola. (x) Trechispora mollusca.
Fig 1. Basidiomata of represented taxa of polypores. (a) Albatrellus piceiphilus. (b) Anomoloma submyceliosum. (c) Aporpium macroporum. (d) Bo-letopsis macrocarpa. (e) Bondarcevomyces taxi. (f) Coltricia perennis. (g) Favolaschia longistipitata. (h) Fistulina hepatica. (i) Fomes fomentarius. (j) Ganoderma sinense. (k) Gloeophyllum sepiarium. (l) Heterobasidion insulare. (m) Hexagonia apiaria. (n) Laetiporus xinjiangensis. (o) Lentinus arcularius. (p) Megasporoporia bannaensis. (q) Phellinus nigricans. (r) Polyporus umbellatus. (s) Porodaedalea himalayensis. (t) Radulotubus resupinatus. (u) Rhodofomes roseus. (v) Sanghuangporus sanghuang. (w) Sistotrema muscicola. (x) Trechispora mollusca.With the advancement of scientific investigations, mycolo-gists are not only describing new taxa of polypores but also focusing on polypore diversity and species composition. In-vestigations have been done in certain mountains, provinces, or countries on the species richness and composition of pol-ypores (Nunez & Ryvarden 1999; Nikitsky & Schigel 2004; Hottola & Siitonen 2008; Kotiranta 2009; Dai 2012; Ryvarden 2018b; Alem et al. 2021; Korhonen et al. 2021; Ma et al. 2022; Wu et al. 2022c; Cui et al. 2025b). Some studies were con-ducted in countries or continents, such as China, Africa, Eu-rope, and North America (Zhou et al. 2016; Ryvarden et al. 2022; Ryvarden & Melo 2014, 2017; Xu et al. 2025). Only a small number of studies have analyzed the species richness and composition of polypores across different continents. Wu et al. (2022a) compiled a total of 1,337 polypores from China, Europe, and North America. Later, Zhao et al. (2024) collated 1,902 polypore species from tropical Africa, Asia, and America. Combining these two studies, a total of 2,670 polypore species was reported, belonging to 282 genera, 53 families, and eleven orders, of Agaricomycetes within Basidiomycota. These taxa were recorded from six zones, including tropical Africa, tropical Asia, tropical America, China, Europe, and North America (Zhao et al. 2024).
Although Zhao et al. (2024) have analyzed the species composition and distribution of polypores in six zones, includ-ing three in temperate zones (China, Europe, and North America), and three in tropical zones (Africa, Asia, and Amer-ica), there were still many areas that were not considered, such as Australia, New Zealand, central and south Asia, Japan, Korea, and central America. In addition, with the advancement of scientific investigations and phylogenetic relationships, many new taxa of polypores have been described based on morphology, phylogeny, and divergence time analyses (Liu et al. 2023b; Zhou et al. 2023; Fryssouli et al. 2024; Hussain et al. 2024a, 2024b; Spirin et al. 2024; Viner et al. 2024; Wang et al. 2024b). For instance, Zhou et al. (2023) proposed two new families, Hirschioporaceae and Trichaptaceae, ten new spe-cies, and 28 new combinations based on 625 specimens from 19 countries. Spirin et al. (2024) re-examined the genus Fom-itopsis, and accepted 128 species according to ITS, ITS + TEF1, ITS + TEF1 + RPB1, and ITS + TEF1 + RPB2 datasets. Wang et al. (2024b) accepted 37 taxa of the genus Phy-sisporinus, consisting of 12 new species based on the phylo-genetic and morphological analyses. Therefore, an updated global checklist of polypores is needed.
Here, the checklist of polypores was obtained from the Fungal Names database (Wang et al. 2023b) and published articles. Then, systematic analyses of the global species richness and distribution patterns of polypores were performed, including species composition, Sørensen similarity index (SS) among different continents, nutritional modes, year published, and scholars' contribution of polypore names. The bibliometric analyses were performed using the Web of Science of Core Collection database.
Table 1. Global species composition of polyporesOrders
(The number of families are in brackets)Families
(The number of genera are in brackets)Genera Number of species Agaricales (8) Cystostereaceae (1) Crustomyces 1 Fistulinaceae (2) Fistulina 10 Pseudofistulina 2 Mycenaceae (3) Favolaschia 109 Filoboletus 17 Panellus 10 Pleurotaceae (1) Resupinatus 8 Porotheleaceae (1) Porotheleum 1 Radulomycetaceae (1) Radulotubus 1 Schizophyllaceae (1) Porodisculus 1 Stephanosporaceae (1) Lindtneria 9 Amylocorticiales (1) Amylocorticiaceae (2) Anomoloma 8 Anomoporia 7 Auriculariales (1) Auriculariaceae (1) Elmerina 12 Incertae sedis (3) Aporpium 3 Heteroradulum 2 Protomerulius 7 Boletales (1) Tapinellaceae (1) Bondarcevomyces 1 Cantharellales (1) Hydnaceae (1) Sistotrema 9 Gloeophyllales (1) Gloeophyllaceae (4) Gloeophyllum 13 Griseoporia 1 Hispidaedalea 1 Osmoporus 2 Hymenochaetales (13) Chaetoporellaceae (2) Echinoporia 3 Kneiffiella 5 Coltriciaceae (1) Coltricia 71 Hirschioporaceae (4) Hirschioporus 8 Nigrohirschioporus 11 Pallidohirschioporus 8 Perennihirschioporus 5 Hymenochaetaceae (40) Arambarria 1 Aurificaria 1 Coniferiporia 4 Cyanotrama 1 Cylindrosporus 1 Flaviporellus 1 Fomitiporella 26 Fomitiporia 73 Fulvifomes 77 Fulvoderma 4 Fuscoporia 106 Hydnoporia 1 Hymenochaete 7 Inocutis 11 Inonotopsis 1 Inonotus 100 Meganotus 1 Mensularia 5 Neomensularia 7 Neophellinus 1 Nothonotus 1 Nothophellinus 1 Ochroporus 2 Ochrosporellus 11 Onnia 10 Pachynotus 1 Perenninotus 1 Phellinidium 5 Phellinopsis 10 Phellinus 37 Phellopilus 1 Phylloporia 96 Polystictus 4 Porodaedalea 20 Pseudoinonotus 8 Pseudophylloporia 1 Pyrrhoderma 8 Rigidonotus 2 Sanghuangporus 20 Tropicoporus 57 Hyphodontiaceae (1) Hyphodontia 2 Neoantrodiellaceae (1) Neoantrodiella 2 Nigrofomitaceae (1) Nigrofomes 3 Rigidoporaceae (4) Bridgeoporus 2 Laetifomes 1 Leucophellinus 2 Rigidoporus 52 Schizoporaceae (3) Paratrichaptum 1 Poriodontia 1 Xylodon 17 Sideraceae (1) Sidera 19 Trichaptaceae (1) Trichaptum 8 Tubulicrinaceae (1) Tubulicrinis 8 Umbellaceae (1) Umbellus 2 Incertae sedis (3) Hastodontia 2 Podocarpioporus 2 Pseudotrichaptum 1 Polyporales (26) Adustoporiaceae (6) Adustoporia 1 Amyloporia 5 Austroporia 1 Lentoporia 2 Resinoporia 12 Rhodonia 6 Auriporiaceae (1) Auriporia 4 Cerrenaceae (5) Cerrena 11 Irpiciporus 7 Lividopora 4 Pseudospongipellis 4 Raduliporus 1 Fibroporiaceae (3) Fibroporia 9 Microporellopsis 1 Pseudofibroporia 1 Fomitopsidaceae (16) Anthoporia 1 Antrodia 35 Buglossoporus 4 Daedalea 15 Eucalyptoporia 1 Fomitopsis 141 Melanoporia 1 Neolentiporus 1 Niveoporofomes 1 Parmastomyces 3 Phaeodaedalea 1 Piptoporus 2 Rhodoantrodia 2 Rubellofomes 2 Spelaeomyces 1 Subantrodia 1 Fragiliporiaceae (1) Fragiliporia 1 Ganodermataceae (14) Amauroderma 60 Amaurodermellus 1 Cristataspora 2 Foraminispora 5 Furtadoella 3 Ganoderma 214 Haddowia 2 Humphreya 3 Magoderna 2 Neoganoderma 1 Sanguinoderma 21 Sinoganoderma 1 Tomophagus 2 Trachydermella 1 Gelatoporiaceae (3) Cinereomyces 4 Obba 3 Sebipora 1 Grifolaceae (2) Aegis 2 Grifola 10 Incrustoporiaceae (3) Gloeoporellus 1 Skeletocutis 94 Tyromyces 127 Irpicaceae (11) Ceriporia 70 Crystallicutis 4 Flavodon 1 Gloeoporus 27 Hydnopolyporus 1 Irpex 42 Leptoporus 4 Meruliopsis 16 Meruliporia 2 Resiniporus 2 Trametopsis 5 Ischnodermataceae (2) Ischnoderma 7 Lasiochlaena 1 Laetiporaceae (6) Kusaghiporia 1 Laetiporus 23 Macrohyporia 3 Pseudophaeolus 2 Wolfiporiella 4 Wolfiporiopsis 1 Laricifomitaceae (3) Gilbertsonia 1 Laricifomes 1 Ryvardenia 2 Meripilaceae (1) Meripilus 4 Meruliaceae (18) Aurantiopileus 4 Aurantiporus 6 Ceriporiopsis 43 Ceriporiopsoides 2 Geesterania 2 Luteoporia 3 Merulius 1 Mycoacia 1 Pappia 2 Phlebia 1 Phlebicolorata 2 Phlebiporia 1 Physisporinus 37 Porpomyces 2 Pseudonadsoniella 1 Pseudophlebia 2 Spongipellis 6 Phaeolaceae (3) Melanoporella 1 Phaeolus 6 Wolfiporia 4 Phanerochaetaceae (9) Bjerkandera 14 Geliporus 1 Hapalopilus 14 Oxychaete 1 Phanerina 1 Phanerochaete 1 Phlebiopsis 1 Pseudohyphodermella 1 Riopa 2 Piptoporellaceae (1) Piptoporellus 5 Podoscyphaceae (2) Abortiporus 5 Heteroporus 1 Polyporaceae (103) Abundisporus 7 Amylosporia 1 Atroporus 3 Aurantioporia 2 Australoporus 1 Cerarioporia 1 Cerioporus 3 Cinereomycetella 1 Citrinoporia 2 Colospora 2 Coriolopsis 22 Coriolus 6 Crassisporus 6 Cryptoporus 3 Cubamyces 3 Cyanoporus 2 Cystidioporia 1 Cystostiptoporus 1 Daedaleopsis 10 Datronia 7 Datroniella 6 Dendroporia 1 Dextrinoporus 1 Dichomitus 15 Donkioporia 2 Donkioporiella 1 Earliella 1 Echinochaete 5 Elfvingia 2 Favolus 33 Flammeopellis 1 Fomes 20 Fomitella 1 Funalia 10 Fuscocerrena 1 Globifomes 1 Grammothele 18 Grammothelopsis 7 Haploporus 29 Hexagonia 24 Hirticrusta 2 Hornodermoporus 1 Jorgewrightia 15 Laccocephalum 2 Leifiporia 2 Lentinus 7 Lenzites 18 Lignosus 7 Loweporus 4 Luteoperenniporia 4 Macroporia 4 Macrosporia 1 Mariorajchenbergia 7 Megasporia 10 Megasporoporia 7 Megasporoporiella 1 Melanoderma 3 Microporellus 20 Microporus 13 Minoporus 1 Mycobonia 1 Navisporus 10 Neodatronia 2 Neodictyopus 3 Neofavolus 9 Neofomitella 4 Neoporia 3 Niveoporia 3 Perenniporia 85 Perenniporiella 5 Perenniporiopsis 3 Phaeotrametes 1 Physisporus 2 Picipes 32 Podofomes 5 Polyporellus 1 Polyporopsis 1 Polyporus 207 Poria 22 Poriella 4 Porogramme 18 Poronidulus 2 Pseudofavolus 4 Pseudogrammothele 1 Pseudomegasporoporia 2 Pseudopiptoporus 2 Pyrofomes 7 Rhizoperenniporia 1 Royoporus 1 Rubroporus 2 Sparsitubus 1 Szczepkamyces 2 Theleporus 10 Thermophymatospora 1 Tinctoporellus 1 Trametes 145 Tropicoporia 4 Truncatoporia 2 Truncospora 12 Vanderbylia 10 Vanderbyliella 1 Xanthoperenniporia 5 Yuchengia 2 Postiaceae (17) Amaropostia 4 Amylocystis 2 Aurantipostia 1 Austropostia 6 Calcipostia 1 Cyanosporus 42 Cystidiopostia 5 Fuscopostia 7 Jahnoporus 4 Nothofagiporus 1 Oligoporus 15 Osteina 5 Postia 24 Ptychogaster 8 Resupinopostia 2 Spongiporus 11 Tenuipostia 1 Pycnoporellaceae (1) Pycnoporellus 2 Sarcoporiaceae (1) Sarcoporia 4 Steccherinaceae (23) Antella 4 Antrodiella 62 Atraporiella 2 Austeria 1 Butyrea 2 Caudicicola 1 Citripora 2 Elaphroporia 1 Flabellophora 9 Flaviporus 14 Frantisekia 3 Junghuhnia 26 Lamelloporus 1 Laschia 2 Loweomyces 4 Metuloidea 3 Mycorrhaphium 1 Niemelaea 3 Nigroporus 7 Rhomboidia 1 Steccherinum 20 Trullella 8 Xanthoporus 2 Taiwanofungaceae (1) Taiwanofungus 2 Incertae sedis (6) Climacocystis 3 Diacanthodes 6 Diplomitoporus 32 Erastia 3 Fabisporus 1 Henningsia 5 Russulales (5) Albatrellaceae (4) Albatrellopsis 3 Albatrellus 35 Byssoporia 1 Polyporoletus 3 Bondarzewiaceae (6) Amylonotus 6 Amylosporus 12 Bondarzewia 15 Heterobasidion 16 Rigidoporopsis 3 Wrightoporia 30 Echinodontiaceae (2) Echinodontium 1 Larssoniporia 2 Hericiaceae (2) Pseudowrightoporia 11 Wrightoporiopsis 6 Terrestriporiaceae (1) Terrestriporia 1 Thelephorales (2) Bankeraceae (1) Boletopsis 11 Thelephoraceae (1) Lenzitopsis 2 Trechisporales (1) Hydnodontaceae (2) Cristelloporia 4 Trechispora 20 -
Checklist collection of polypores
In this study, the polypore species were downloaded from the Fungal Names database (Wang et al. 2023b, https://nmdc.cn/fungalnames/, accessed on 1 November 2024), and then verified by consulting scientific literature and other databases, including MycoBank (https://www.mycobank.org), Index Fungorum (https://www.indexfungorum.org), and Global Biodiversity Information Facility (GBIF, https://www.gbif.org). In addition, new taxa were drawn from publications in May 2025 (Bashir et al. 2024; Bittencourt et al. 2024a, 2024b; Cui et al. 2024; de Targino et al. 2024; Dong et al. 2024; Fryssouli et al. 2024; Gunaseelan et al. 2024; He et al. 2024b; Hussain et al. 2024a, 2024b, 2025; Ji et al. 2024; Luo et al. 2024a; Liu et al. 2024a, 2024b, 2025a; Niu et al. 2024; Saha et al. 2024a, 2024b; Salvador-Montoya et al. 2024; Suh et al. 2024; Spirin et al. 2024; Tang et al. 2024; Umar et al. 2024; Viner et al. 2024; Wang et al. 2024a, 2024b, 2024c, 2024d; Wei et al. 2024a, 2024b; Wu et al. 2024a; Xu et al. 2024; Yuan & Zhao 2024; Zabin et al. 2024; Zhang et al. 2024a, 2024b, 2024c, 2024d, 2025; Zheng et al. 2024; Zhou et al. 2024a, 2024b; Zhu et al. 2024; Choudhary & Uniyal 2025; Jérusalem et al. 2025; Li et al. 2025; Oliveira et al. 2025; Shao et al. 2025; Tian et al. 2025; Yayoro et al. 2025; Zhang & Luo 2025). The checklists of polypores consisted of taxonomic ranks, current name and basionym name of each species, type of rot, reference se-quences (mainly ITS or LSU), distribution, type locality (coun-try), year of publication of current name and basionym name, and references (File S1).
-
Classification of polypores
Polypores were mostly defined as wood-inhabiting basidio-mycetes with a poroid hymenophore, traditionally, polypores belonged to the Aphyllophorales (Fig. 1). So, some species with a hymenophore configuration other than poroid are also included, for instance, some species with lamellae or sublam-ellate, irpicoid, hydnoid, and corticioid hymenophore (Ryvarden 1991; Wu et al. 2022a; Zhao et al. 2024). Currently, polypores belong to 11 orders within the Agaricomycetes, namely Agaricales, Amylocorticiales, Auriculariales, Boletales, Can-tharellales, Gloeophyllales, Hymenochaetales, Polyporales, Russulales, Thelephorales, and Trechisporales (Ryvarden 1991; Zhao et al. 2015; Wu et al. 2022a; Zhao et al. 2024). In addition, a classification hierarchy of genus and family was generated with Lifemap (https://lifemap-ncbi.univ-lyon1.fr/, 20th July 2025) using the checklist of polypores (File S1), to compare with the previous studies (Zhou et al. 2023; Hyde et al. 2024b; Xu et al. 2025; Zhao et al. 2025a), a diagram was generated with iTOL (https://itol.embl.de/) with the number of species in each genus.
-
Nutritional modes
Nutritional modes of polypores were divided into four types, including white rot, brown rot, ectomycorrhizal, and uncertain (Wu et al. 2022a; Zhao et al. 2024). It was noted that some species of Coltricia have been identified as ectomycorrhizal, including C. abieticola, C. dependens, C. perennis, and C. weii, while many others were treated as uncertain (Tedersoo et al. 2007; Wu et al. 2022b; Zhao et al. 2023b).
-
Distribution patterns of polypores
The distribution was also divided into four zones, namely bo-real, temperate, subtropical, and tropical. The geographical distribution included six continents, viz., Africa, Asia, Europe, North America, Oceania, and South America.
-
Bibliometric analyses
To obtain the trends in research concerned with polypores, the bibliometric analyses were performed using the search keywords “polypores”, “Hymenochaetaceae”, and “Polypo-raceae” in the “Web of Science Core Collection” database for 2000–2024. The species numbers of the families Hymenochaetaceae and Polyporaceae accounted for ap-proximately 60 % of the polypores according to Zhao et al. (2024), so these keywords were added in this study. We se-lected “tab defined file format” to export author, title, source, abstract, and other information, the co-occurrence network and research hotspot plots were visualized using VOSviewer 1.6.20 (Arruda et al. 2022).
-
Data analyses
The Sørensen Similarity index (SS) among the studied continents was tested using the following equation: SS=2C/(A+B), where A and B were the species numbers in the two compared continents, and C was the number of species recorded in both continents (Sørensen 1948; Wu et al. 2022a; Zhao et al. 2024). Columnar and line charts, and Voronoi diagrams were performed using the R programs ggplot2 (Wilkinson 2011), and voronoiTreemap (https://github.com/uRosConf/voronoiTreemap, accessed on 10th February 2024), respectively, in the R package v4.1.2 (R Core Team 2013). Flower plots were tested using the imageGP 2 website (Chen et al. 2024; http://www.bic.ac.cn/BIC/).
-
Global species composition of polypores
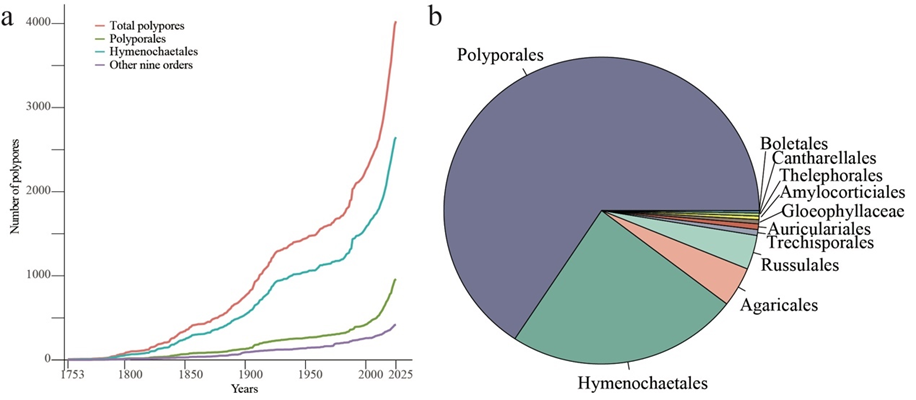
Since Linnaeus named polypore species using binomial no-menclature in 1753, the number of polypore species has steadily increased. By 1912, after 159 years, the number of polypores exceeded 1,000, reaching 1,009. By 1989, 77 years later, it surpassed 2,000, reaching 2,031. By 2016, it exceeded 3,000 in just 27 years, reaching 3,010, and more than 1,000 new species have been described from 2017 to March 2025. Currently, 4,026 species have been described worldwide until May 2025 (Fig. 2, Fig. 3a, and File S1). Meanwhile, the pol-ypore numbers in the main orders, Polyporales and Hy-menochaetales, show similar trends.
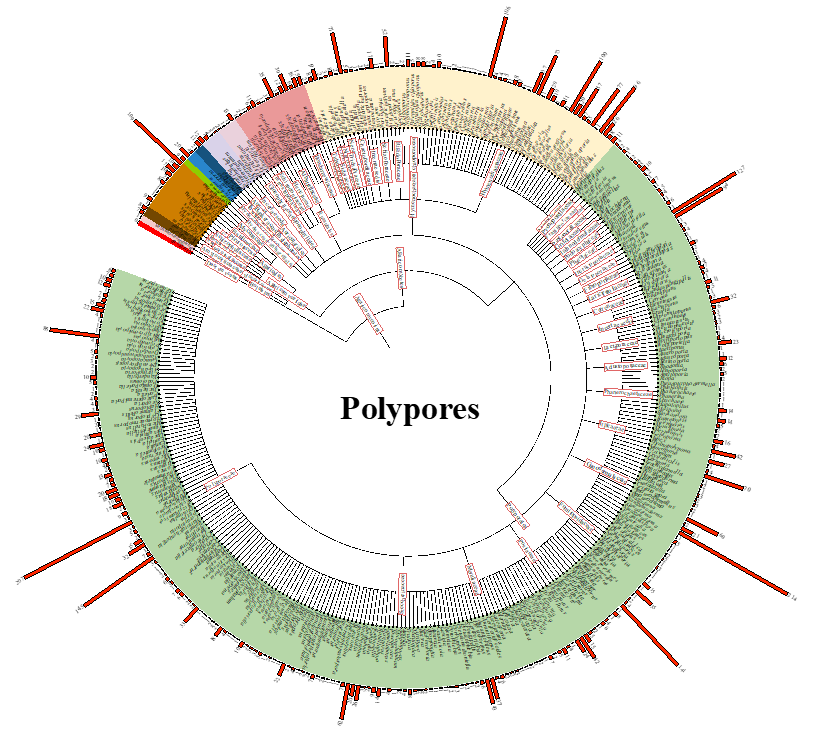 Fig 2. Diagram showing the global species richness of polypores, consisting of 4,026 species, 368 genera, 60 families, and 11 orders within the class Agaricomycetes. The red bars on the outer circle indicate the number of species within each corresponding genus. The colours on the inner circle represent the different orders.
Fig 2. Diagram showing the global species richness of polypores, consisting of 4,026 species, 368 genera, 60 families, and 11 orders within the class Agaricomycetes. The red bars on the outer circle indicate the number of species within each corresponding genus. The colours on the inner circle represent the different orders.In this study, a total of 4,026 polypores were collated worldwide, belonging to 11 orders, 60 families, and 368 genera within the class Agaricomycetes (Fig. 3b, Table 1, and File S1). Among these species, the order Polyporales has 2,648 species, accounting for 65.77 %, in 26 families and 262 genera (Table 2). The order Hymenochaetales has 961 species, accounting for 23.87 %, in 13 families and 64 genera. The order Agaricales has 169 species, accounting for 4.20 %, in eight families and 11 genera. The order Russulales has 145 species, accounting for 3.60 %, in five families and 15 genera. The order Auricu-lariales has 24 species, accounting for 0.60 %, in one family and four genera. The order Trechisporales has 24 species, accounting for 0.60 %, in one family and two genera. The order Gloeophyllaceae has 17 species, accounting for 0.42 %, in one family and four genera. The order Amylocorticiales has 15 species, accounting for 0.38 %, in one family and two genera. The order Thelephorales has 13 species, accounting for 0.32 %, in two families and two genera. The order Can-tharellales has nine species, accounting for 0.22 %, in one family and one genus. The order Boletales has one species, accounting for 0.02%, in one family and one genus.
At the family level, Polyporaceae (Polyporales), Hy-menochaetaceae (Hymenochaetales), and Steccherinaceae (Polyporales) contained the greatest number of genera, with 103, 40, and 23, respectively and Polyporaceae, Hy-menochaetaceae, and Ganodermataceae (Polyporales) had the most species, with 1,170, 725, and 318 species, respectively. At the genus level, there were eight genera that consisted of more than 100 species (Fig. 2, Table 1), namely Favolaschia (109 species), Fomitopsis (141 species), Fuscoporia (106 species), Ganoderma (214 species), Inonotus (100 species), Polyporus (207 species), Trametes (145 species), and Tyromyces (127 species).
Table 2. A composition and proportion of 11 orders with polyporesOrders Number of families Number of genera Number of species Proportion of total polypores Polyporales 26 262 2,648 65.77% Hymenochaetales 13 64 961 23.87% Russulales 5 15 145 3.60% Auriculariales 1 4 24 0.60% Trechisporales 1 2 24 0.60% Gloeophyllaceae 1 4 17 0.42% Amylocorticiales 1 2 15 0.38% Thelephorales 2 2 13 0.32% Cantharellales 1 1 9 0.22% Boletales 1 1 1 0.02% In addition, we compiled the locality data for type materials of all polypore species, which were distributed in 154 countries or regions (File S2). Among the top 20 countries with type materials, there are a maximum of seven Asian countries and at least one African country (Table 3), totalling 2,815 species found in these regions, which accounts for 69.92 %. Moreover, 2,285 polypores had reference sequences, accounting for 56.76 %, while sequences of 1,741 species are not available so far, accounting for 43.24 % (File S1).
Table 3. Types of polypores in the top 20 countriesRanks Countries Number of types Continents Ranks Countries Number of types Continents 1 China 803 Asia 11 Costa Rica 69 North America 2 USA 315 North America 12 Venezuela 68 South America 3 Brazil 312 South America 13 Argentina 64 South America 4 Malaysia 179 Asia 14 Cuba 64 North America 5 Australia 138 Oceania 15 New Zealand 63 Oceania 6 Indonesia 107 Asia 16 Philippines 62 Asia 7 Japan 104 Asia 17 Cameroon 60 Africa 8 India 101 Asia 18 Sri Lanka 48 Asia 9 Russia 77 Europe 19 Sweden 48 Europe 10 France 72 Europe 20 Finland 47 Europe Note: All countries with taxonomic types in polypores are listed in File S2. -
The highest species richness of Asia
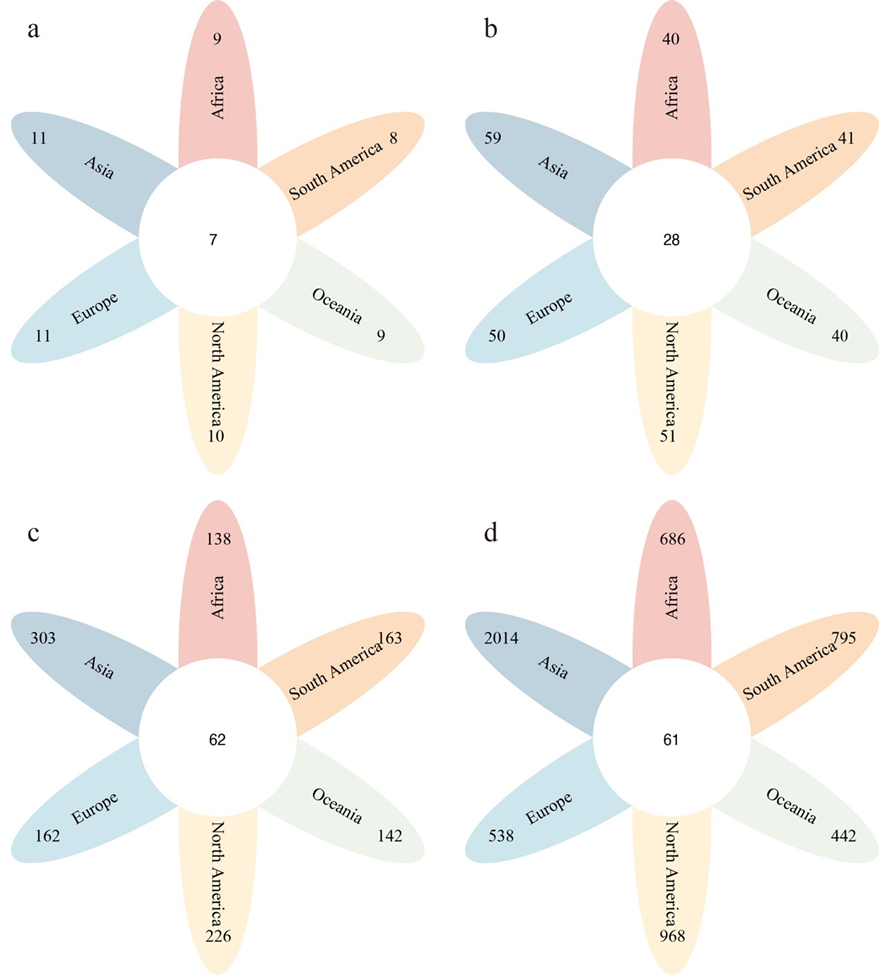
Among 4,026 polypores across six continents, Asia had the highest number of species with 2,014 species from 11 orders, 59 families, and 303 genera (Fig. 4 and Table 4). North Amer-ica followed with 968 species distributed among ten orders, 51 families, and 226 genera. South America ranked third with 795 species in eight orders, 41 families, and 163 genera. Africa came fourth with 686 species, belonging to nine orders, 40 families, and 138 genera. Europe has 538 species from 11 orders, 50 families, and 162 genera, and Oceania has the fewest, with 442 species in nine orders, 40 families, and 142 genera.
Currently, there are 11 orders of polypores, with seven of them found in the six continents, viz., Agaricales, Auriculariales, Gloeophyllales, Hymenochaetales, Polyporales, Russulales, and Trechisporales (Fig. 4a), accounting for 63.6 %. Only Asia and Europe contain all 11 orders, and South America has the fewest, with nine orders. At the family level, a total of 60 fami-lies was collected, with 28 families found in the six continents, comprising 46.7 % (Fig. 4b). Asia, North America, and Europe (mainly located in the Northern Hemisphere) have a higher number of families, with counts of 59, 51, and 50, respectively, compared to South America, Africa, and Oceania (mainly lo-cated in the Southern Hemisphere), which have 41, 40, and 40 families, respectively. Regarding genera, there were 368 gen-era of polypores, with 62 shared genera found in the six con-tinents, making up 16.8 % (Fig. 4c). Asia and North America have the largest number of genera. In addition, there are only 61 shared species found in the six continents, which repre-sents only 1.5 % of all polypores, and Asia has the highest species richness among all six continents (Fig. 4d).
Table 4. Composition of polypores in Africa, Asia, Europe, North America, Oceania, and South AmericaOrders Families (Abbreviation) Number of genera Number of species Africa Asia Europe North America Oceania South America Africa Asia Europe North America Oceania South America Agaricales Cystostereaceae (Cy) 0 1 0 0 0 0 0 1 0 0 0 0 Fistulinaceae (Fi) 1 2 1 2 1 2 1 4 1 4 3 5 Mycenaceae (My) 2 3 1 3 3 3 22 56 1 23 17 51 Pleurotaceae (Pl) 0 1 1 1 1 0 0 3 2 4 2 0 Porotheleaceae (Po) 0 1 1 1 0 0 0 1 1 1 0 0 Radulomycetaceae (Ra) 0 1 0 0 0 0 0 1 0 0 0 0 Schizophyllaceae (Sc) 1 0 0 1 1 1 1 0 0 1 1 1 Stephanosporaceae (St) 1 1 1 1 0 0 1 1 5 6 0 0 Amylocorticiales Amylocorticiaceae (Am) 1 2 2 2 0 1 1 11 5 6 0 1 Auriculariales Auriculariaceae (Au) 1 1 1 1 1 1 1 9 1 2 3 3 Incertae sedis (In) 1 3 2 1 1 1 3 6 2 3 1 2 Boletales Tapinellaceae (Ta) 0 1 1 0 0 0 0 1 1 0 0 0 Cantharellales Hydnaceae (Hy) 0 1 1 1 1 0 0 5 8 3 1 0 Gloeophyllales Gloeophyllaceae (Gl) 1 4 1 2 1 1 4 14 7 8 6 3 Hymenochaetales Chaetoporellaceae (Ch) 1 2 1 2 0 2 1 5 1 2 0 3 Coltriciaceae (Co) 1 1 1 1 1 1 6 47 5 11 5 11 Hirschioporaceae (Hi) 1 4 2 4 2 2 1 20 4 9 3 6 Hymenochaetaceae (Hm) 13 31 18 27 15 19 73 377 54 181 57 123 Hyphodontiaceae (Hp) 1 1 1 1 1 1 1 1 1 1 2 1 Incertae sedis (In) 0 3 2 2 1 0 0 4 3 2 1 0 Neoantrodiellaceae (Ne) 0 1 0 0 0 0 0 2 0 0 0 0 Nigrofomitaceae (Ni) 1 1 1 1 1 1 1 3 1 1 1 1 Rigidoporaceae (Ri) 2 3 1 2 3 1 15 35 8 16 12 18 Schizoporaceae (Sh) 1 3 1 1 1 1 3 16 4 5 5 6 Sideraceae (Si) 0 1 1 1 1 1 0 14 3 3 5 2 Trichaptaceae (Tr) 1 1 0 1 1 1 1 4 0 4 1 2 Tubulicrinaceae (Tu) 1 1 1 1 0 1 1 5 6 3 0 1 Umbellaceae (Um) 0 1 1 1 1 0 0 1 1 1 1 0 Polyporales Adustoporiaceae (Ad) 1 5 4 4 0 2 1 17 7 12 0 2 Auriporiaceae (Ar) 0 1 1 1 0 1 0 3 1 1 0 1 Cerrenaceae (Ce) 4 4 3 5 3 3 8 11 4 13 5 11 Fibroporiaceae (Fb) 2 2 1 1 1 1 2 7 6 3 1 1 Fomitopsidaceae (Fo) 3 13 5 4 5 4 24 98 49 56 29 24 Fragiliporiaceae (Fr) 0 1 0 0 0 0 0 1 0 0 0 0 Ganodermataceae (Ga) 7 10 1 6 3 9 97 137 11 47 22 71 Gelatoporiaceae (Ge) 0 3 2 2 1 2 0 5 2 2 1 3 Grifolaceae (Gr) 0 1 1 1 1 2 0 4 1 1 3 5 Incertae sedis (In) 3 5 4 5 3 4 20 10 8 10 3 16 Incrustoporiaceae (Ic) 2 2 2 2 3 2 41 94 37 42 31 23 Irpicaceae (Ir) 6 9 9 11 7 6 20 83 41 50 25 36 Ischnodermataceae (Is) 1 1 2 1 1 1 1 2 2 1 2 2 Laetiporaceae (La) 3 4 2 4 2 1 6 16 4 12 2 3 Laricifomitaceae (Lr) 0 1 1 2 1 0 0 1 1 2 2 0 Meripilaceae (Me) 0 1 1 1 1 0 0 2 1 2 1 0 Meruliaceae (Mr) 3 15 7 10 4 9 27 51 21 31 6 22 Phaeolaceae (Ph) 2 2 1 2 1 1 2 7 1 3 1 1 Phanerochaetaceae (Pa) 3 9 3 2 3 2 6 19 8 12 4 9 Piptoporellaceae (Pi) 1 1 1 0 0 0 1 4 1 0 0 0 Podoscyphaceae (Pd) 1 1 1 1 2 1 2 2 2 2 2 2 Polyporaceae (Py) 44 88 30 54 37 48 225 517 91 232 128 249 Postiaceae (Ps) 4 14 14 13 11 4 6 89 42 34 20 8 Pycnoporellaceae (Pc) 0 1 1 1 0 0 0 1 1 1 0 0 Sarcoporiaceae (Sa) 0 1 1 1 0 1 0 2 2 3 0 1 Steccherinaceae (Se) 5 15 12 16 8 12 32 75 39 43 16 42 Taiwanofungaceae (Ta) 0 1 0 0 0 0 0 2 0 0 0 0 Russulales Albatrellaceae (Al) 2 2 3 4 0 0 3 30 7 17 0 0 Bondarzewiaceae (Bo) 3 6 2 4 3 3 12 47 5 14 6 12 Echinodontiaceae (Ec) 1 1 0 2 0 1 1 1 0 2 0 1 Hericiaceae (He) 2 2 0 2 1 1 3 10 0 3 1 2 Terrestriporiaceae (Te) 0 1 0 0 0 0 0 1 0 0 0 0 Thelephorales Bankeraceae (Ba) 1 1 1 1 1 0 1 5 5 5 1 0 Thelephoraceae (Th) 0 1 1 0 0 0 0 1 1 0 0 0 Trechisporales Hydnodontaceae (Hd) 2 2 1 2 1 1 8 12 12 12 3 8 The Sørensen similarity index (SS) values were also cal-culated for six continents (Table 5). Europe and North America had the highest SS values, indicating greater species similarity. North and South America followed closely in similarity. Africa and Asia exhibited the highest species similarities with Oce-ania and Europe, respectively, while the lowest SS value was observed between Asia and South America.
Table 5. Similarity matrix among continentsContinents Africa Asia Europe North-America Oceania South-America Africa 0.1519 0.1471 0.2019 0.2358 0.1999 Asia 0.1519 0.2322 0.2200 0.1417 0.1089 Europe 0.1471 0.2322 0.3692 0.1694 0.1185 North-America 0.2019 0.2200 0.3692 0.1858 0.3358 Oceania 0.2358 0.1417 0.1694 0.1858 0.1956 South-America 0.1999 0.1089 0.1185 0.3358 0.1956 -
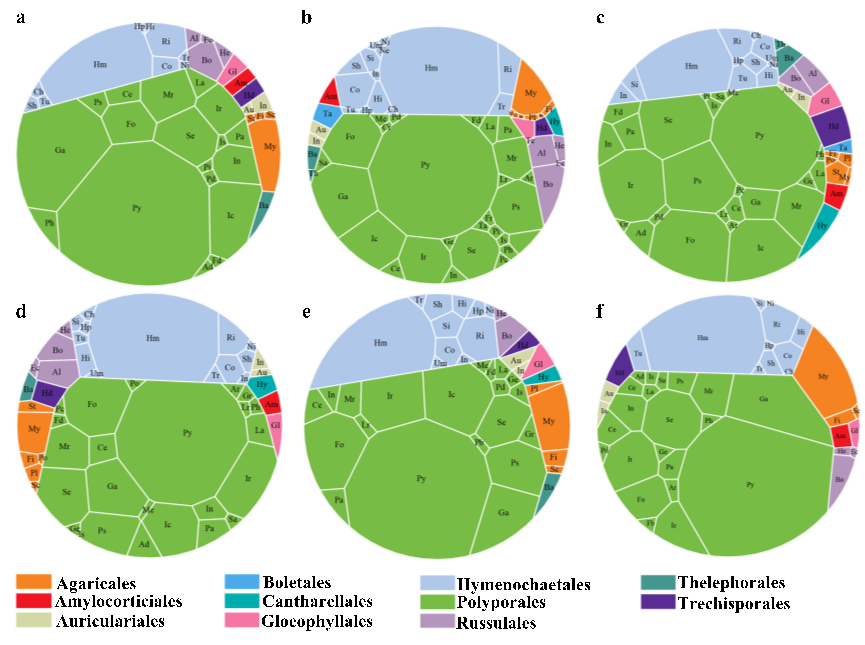
Polyporales and Hymenochaetales dominated six continents
The order Polyporales dominated across six continents, with proportions ranging from 62.5% in Asia and 75.95% in Africa (Fig. 5). Next was the Hymenochaetales, comprising 15.01 % in Africa to 26.51 % in Asia. Agaricales ranked third, with a range of 1.86 % in Europe to 7.17 % in South America. Russulales followed in fourth, showing proportions from 1.61 % in Oceania to 4.5 % in Asia. Additionally, eight other orders collectively accounted for only 4.02 % in South America to 10.04 % in Europe.
At the family level, Polyporaceae of Polyporales was prev-alent across six continents, ranging from 16.91 % in Europe to 32.80 % in Africa. Hymenochaetaceae from Hymenochaetales followed, making up 10.64 % in Africa and 18.72 % in Asia. In addition, some families within Polyporales, such as Fomitop-sidaceae, Ganodermataceae, Incrustoporiaceae, Irpicaceae, and Meruliaceae, as well as Mycenaceae from Agaricales also contributed significantly to the overall proportions of polypores.
-
Polypore distribution in four climate zones
In this study, four climate zones were classified, viz., boreal, temperate, subtropical, and tropical. The distribution of pol-ypores is listed in File S1. The temperate zone has the highest richness of order, family, and genus with 11, 54, 257, respec-tively, compared to the boreal zone (10 orders, 33 families, and 88 genera), the subtropical zone (9 orders, 46 families, and 214 genera), and the tropical zone (9 orders, 48 families, and 238 genera, Fig. 6a-c). At the species level, the tropical and tem-perate zones have the greatest number, with 2,058 and 1,518 species, respectively, followed by the subtropical zone with 908 species and the boreal zone with 174 species (Fig. 6d).
On the other hand, the distribution of polypore species across four climate zones on six continents reveals three distinct trends (Fig. S1). In Africa and South America, the number of species increased from the boreal zone to the tropical zone, with the highest species richness in the tropical zones. In Asia, North America, and Oceania, the number of species initially increased, then decreased, and increased again, with the highest species richness in the temperate or tropical zones. In Europe, the trend shows an initial increase followed by a de-crease from the boreal to the subtropical zones, with the highest species richness found in the temperate zone.
-
The relationships between nutritional modes and climate zones
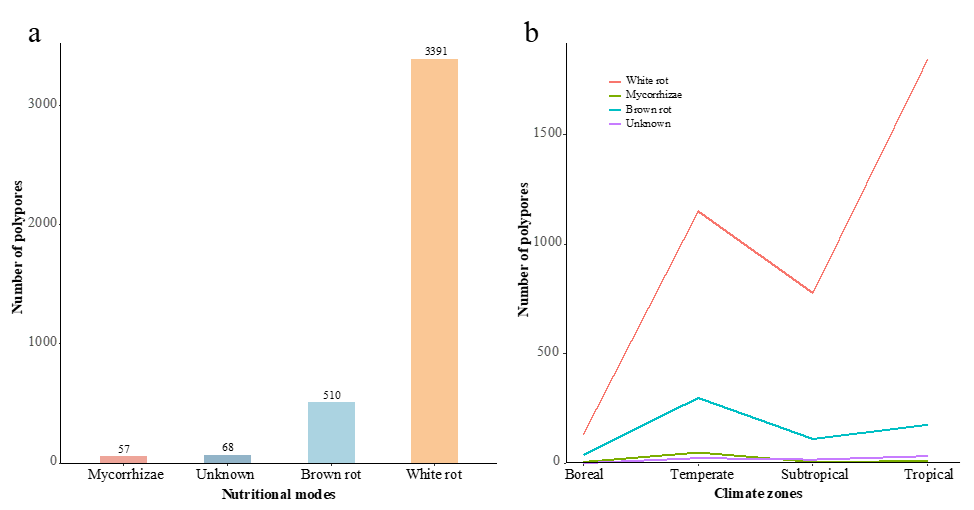
Four nutritional modes were identified, including white rot, brown rot, mycorrhizae, and unknown. Of the 4,026 species, the majority were white rot fungi, comprising 3,391 species, or 84.22 % of the total polypores (Fig. 7a). Brown rot fungi in-cluded 510 species, making up 12.67 % of the total polypores (Fig. 7a). Mycorrhizal fungi and those treated as unknown accounted for a smaller number, with 57 and 68 species, respectively, rep-resenting 1.42 % and 1.69 % of the total polypores (Fig. 7a).
White rot polypores were mainly located in the tropical zone followed by the temperate and subtropical zones (Fig. 7b). In contrast, brown rot polypores predominantly occurred in the temperate zone, with fewer species in the tropical and sub-tropical zones (Fig. 7b). The majority of mycorrhizal fungi were found in the temperate zone (Fig. 7b). Furthermore, white rot fungi were the most prevalent across all six continents, fol-lowed by brown rot fungi, while mycorrhizal fungi were the least common (Fig. S2).
-
Research trends in polypores
The search query obtained 1,148 articles from the “Web of Science Core Collection” database during the 24 years (2000–2024) using three keywords, viz., polypores, Hymenochaeta-ceae, and Polyporaceae. From these articles, 91 unique keywords were identified (File S3), with 30 appearing more than 100 times. Notably, nine terms, activity, basidiomata, China, compound, forest, hyphae, new species, phylogenetic analysis, and species richness, each exceeded 400 occur-rences.
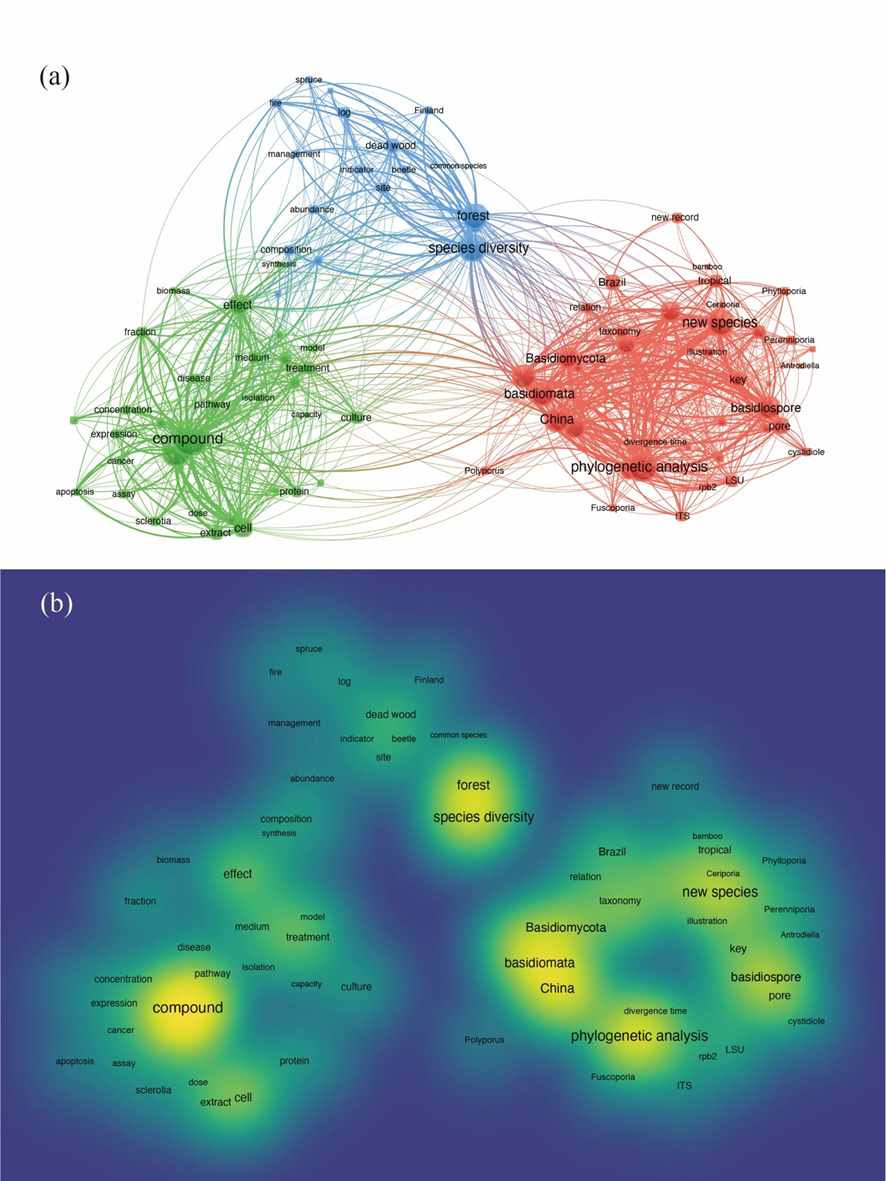 Fig 8. (a) Co-occurrence network plot and (b) research hotspot map using keywords “polypores”, “Hymenochaetaceae”, and “Polyporaceae” based on the Web of Science Core Collection Citation database. The node size represents the frequency of occurrence, and the connecting lines between nodes represent the co-occurrence relationship. Centrality represents the frequency of linkages between different clusters, whereas density represents the frequency of internal links within a cluster.
Fig 8. (a) Co-occurrence network plot and (b) research hotspot map using keywords “polypores”, “Hymenochaetaceae”, and “Polyporaceae” based on the Web of Science Core Collection Citation database. The node size represents the frequency of occurrence, and the connecting lines between nodes represent the co-occurrence relationship. Centrality represents the frequency of linkages between different clusters, whereas density represents the frequency of internal links within a cluster.Co-occurrence network analysis of the keywords revealed three distinct clusters (Fig. 8a). Cluster 1, containing 41 key-words centred around “phylogenetic analyses” and “new spe-cies”, was linked to terms such as Basidiomycota, China, genus, and morphological characters, indicating a focus on the taxonomy of polypores based on morphological and mo-lecular phylogenetic studies. Cluster 2 comprised 32 keywords, with “activity” and “compound” as the core, connected to terms like disease, effect, rat, and treatment. This cluster suggests research related to the applications of polypores. Cluster 3 included 18 keywords, with “forest” and “species diversity” at its centre, linked to factors such as amount, fire, and habitat, highlighting the ecological relationships involving polypores. Furthermore, the research hotspot map corroborated these three clusters as key trends in polypore studies (Fig. 8b).
-
Since the start of the 21st century, the use of molecular biology in fungal taxonomy has led to a significant increase in the number of polypores, a key group within Basidiomycota. Since 2000 to May 2025, 1,808 new polypores have been described, making up 43.54 % of the total according to this study. Notably, since 2018, more than 100 new species have been described almost each year (File S1). Although there were many articles and books decribling new taxa of polypore, some aspects needed futher development, such as global polypore composition and distribution, as well as ge-nomics and ecological. Thus, we focused on the global species richness and distribution patterns of polypores, provide an overview of research trends, and discussed potential issues.
Morphological evidence has long been crucial in fungal taxonomy, but advances in molecular biology over the past 20 years have significantly transformed traditional research methods. The polypores were traditionally treated in the order Aphyllophorales, but now are divided into 11 orders, with new families, genera, and species proposed using molecular analysis (Wu et al. 2022a, 2022b; Ji et al. 2023; Liu et al. 2023a, 2023c; Zhou et al. 2023; Hussain et al. 2024b; Spirin et al. 2024; Spirin & Ryvarden 2024; Zhao et al. 2025b, 2025c). In addition, phylogenetic analyses have revealed cryptic species, further enhancing our understanding of polypore diversity. For in-stance, Korhonen et al. (2018) recognized 11 species from the Skeletocutis nivea species complex based on nuclear ribo-somal internal transcribed spacer (ITS) and translation elon-gation factor TEF1 sequences. Another example is that of one of the most common polypores, Fomes fomentarius. Numerous specimens previously classified as Fomes fomentarius, have been shown to include three distinct species, viz., F. fomentarius, F. heng-duanensis, and F. inzengae, based on ecological, morpholog-ical, phylogenetic, and physiological characteristics (Peintner et al. 2019; Tomšovský et al. 2023; Cui et al. 2024). Cui et al. (2025a) studied the species diversity of the phytopathogenic fungal genus Phaeolus based on phylogeny, divergence times, and biogeography. However, many polypores still lack molec-ular data, particularly type materials, posing a significant challenge to accurately assessing species diversity, as some cannot be reliably identified through morphology alone. Alt-hough some widely distributed species may be comprised of several cryptic species based on molecular evidence, and some species lacking molecular markers may be a synonym of an existing species, we were not willing to discuss these re-lated issues because insufficient evidence supports such species. About 43 % of known polypore species currently are without reference DNA sequences, and even recently, some new species have been proposed based on morphological characteristics only, such as Diplomitoporus and Tyromyces (Hjortstam & Ryvarden 2009; Ryvarden 2018a, 2018b, 2020,2024a, 2024b; Tsigaing et al. 2020; Decock et al. 2021a, 2021b). Hence, the ongoing enrichment of molecular sequence datasets, especially in type specimens or voucher materials, is essential for resolving taxonomic uncertainties and promoting a more robust and comprehensive understanding of fungal diversity.
Many mycologists have collected and studied polypores worldwide, but numerous species remain to be discovered due to factors such as geographical isolation, funding limitations, and the challenges of accessing harsh environments (Zhao et al. 2024). This study finds that Asia has the highest species number of polypores compared to the other five continents, particularly in contrast to Africa, and Central and South Amer-ica. Fortunately, some new polypore species have been de-scribed in countries that were previously under-researched, such as India (Crous et al. 2021; Jayawardena et al. 2022; Senanayake et al. 2023; Gunaseelan et al. 2024), Kenya (Ryvarden 2018a; Decock et al. 2021a; Ryvarden 2023; Jérusalem et al. 2025), Mexico (Valenzuela et al. 2023; Suh et al. 2024), Pakistan (Ahmed et al. 2023; Bashir et al. 2024; Gafforov et al. 2025; Hussain et al. 2024a, 2024b, 2025), and South Korea (Tibpromma et al. 2017; Jung et al. 2018; Cho et al. 2023, 2024), which promoted our understanding of species richness and distribution patterns of polypores across six continents. Moreover, the new taxa of polypores are typically described based on morphology and phylogeny, such as new species within Ceriporia, Coltricia, Fomitiporia, Fulvifomes, Ganoderma, Meripilus, and Nigroporus (Chen et al. 2025a, 2025b; Li et al. 2025; Kumar et al. 2025; Olou et al. 2025; Wang et al. 2025a; Zhao et al. 2025b).
It is important to note that the climate zones and geo-graphical distribution of polypores may be incomplete due to limited research on the species numbers in certain areas and a lack of comprehensive literature records. Previous studies indicated that the temperate zone has the highest number within the class Agaricomycetes, while the latitudinal diversity gradient (LDG) hypothesis suggests that tropical regions har-bour the greatest species diversity of many groups, such as plants and animals (Mittelbach et al. 2007; Varga et al. 2019). Our earlier research (Zhao et al. 2024) also indicated that the tropical zone has the highest species number of polypores. While the lower species number in the subtropical zone compared to the temperate zone suggested that there may be a large number of species in this area it has not yet been investigated.
Studying the species richness of polypores may promote the development and utilization of biological resources. There has been increasing interest in the medicinal properties of polypores, as highlighted by bibliometric analyses that under-score their importance in traditional medicines. For instance, Fomes fomentarius and Inonotus obliquus are recognized as valuable medicinal polypores in European tradition (Grienke et al. 2014; Peintner et al. 2019; Szychowski et al. 2021), and species from the genera Sanghuangporus and Ganoderma are entrenched in traditional Chinese medicine (Ren et al. 2021; Sun et al. 2022a, 2022b; Lin et al. 2023; Wang et al. 2023c; Wu et al. 2024a). Currently, many polypores have been proven to have medicinal properties (Sangdee et al. 2017; Campi et al. 2021; Szychowski et al. 2021; Chafouz et al. 2023; Cheng et al. 2023; Hua et al. 2023; Ghobad-Nejhad et al. 2024; Jiang et al. 2024; Luo et al. 2024b; Shen et al. 2024), indicating further exploration is needed. It is worth noting that in recent years, more than 20 species of polypores have been successfully cultivated in China (Dai 2022).
Most polypores, as wood-decaying fungi, are closely associ-ated with their host trees and play an important role in the stability and regeneration of forest ecosystems (Palviainen et al. 2010; Berglund et al. 2011; Vainio et al. 2011; Zhou & Dai 2012; Krah et al. 2018; Dai et al. 2021; Huang et al. 2022). In this study, the results of co-occurrence network analysis showed that the keyword “forest” and “species diversity” formed another grouping at core, strongly emphasizing signif-icant relationship with polypores. However, due to the impact of human activities, such as forestry practices, polypores are facing severe challenges. Forestry affects biodiversity in vari-ous interconnected ways across multiple temporal and spatial scales (Asplund et al. 2024). The reduction in structural and functional diversity caused by for-estry limits the diversity of habitats and substrates, negatively impacting biodiversity by providing fewer ecological niches for wood-decaying fungi (Sippola et al. 2001; Ranius et al. 2019; Moor et al. 2021; Asplund et al. 2024). On the other hand, the restoration of richness of deadwood in forests can increase the species diversity and populations of wood-decaying fungi (Pasanen et al. 2014; Zibold et al. 2024). Therefore, the conservation of forest ecosystems plays a vital role in maintaining the diversity of polypores.
In recent years, the advancement of multiomics studies on polypores has steadily progressed, which holds great promise for the discovery, development, and application of polypore resources. For example, comparative genomics showed that white-rot and brown-rot fungi had significant distinctions in carbohydrate-active enzyme (CAZyme) gene families, partic-ularly in class II lignin-modifying peroxidase (POD) genes (Floudas et al. 2012; Kohler et al. 2015; Krah et al. 2018). Combining genomics, transcriptomics, and metabolomics to uncover the biosynthetic pathways of secondary metabolites in medicinal polypores has become a major research focus in recent years, especially in the genera Ganoderma and Sanghuangporus (Sun et al. 2022b; Cho et al. 2025; Wang et al. 2025b). Moreover, many studies focused on ecological diver-sification, phylogenomics, mitochondrial genome, and the pathogenic mechanisms of polypore pathogens using multi-omics (Chung et al. 2017; Zhao et al. 2023b, 2025a; Feng et al. 2024; Garcia et al. 2024; Liu et al. 2025b; Ma et al. 2025; Nie et al. 2025). These studies of polypores may be lagging behind other fungal groups, such as ectomycorrhizae and yeasts, not only in the number of published genomes, but, more im-portantly, in study depth (Martin et al. 2016; Miyauchi et al. 2020; Rossi et al. 2021; Sahu et al. 2023; Deng et al. 2025; Mondo & Grigoriev 2025; Qu et al. 2025; Tremble et al. 2024; Yan et al. 2025). Hence, comprehensive multiomics investiga-tions on polypores should be regarded as a long-term research priority, requiring ongoing commitment and innovation.
-
In this study, we present a comprehensive global polypore richness and distribution based on an extensive compilation of database and literature records. A total of 4,026 accepted species belonging to 11 orders, 60 families, and 368 genera within Agaricomycetes highlights the remarkable species richness of polypores and their uneven taxonomic composition, laying a solid foundation for future study on their evolutionary origins and resource utilization.
-
The research is supported by the National Natural Science Foundation of China (Project No. U23A20142 and 32161143013), Hainan Province Science and Technology Special Fund (Project No. ZDYF2023RDYL01), the Hainan Institute of National Park (Project No. HINP, KY-24ZK02), and Young Elite Scientists Sponsorship Program by CAST (Project No. 2023QNRC001).
-
Conceptualization, Zhao H., Yuan H.-S., Dai Y.-C., and Yuan Y.; methodology, Zhao H., Cui Y.-J., and Wang K.; formal analysis, Zhao H. and Cui Y.-J.; resources, Yuan H.-S., Wu F., Dai Y.-C., and Yuan Y.; data curation, Zhao H.; writing—original draft preparation, Zhao H.; writing—review and editing, Yuan H.-S., Wu F., Dai Y.-C., and Yuan Y.; supervision, Dai Y.-C.; project administration, Yuan Y.; funding acquisition, Dai Y.-C., and Yuan Y. All authors have read and agreed to the published version of the manuscript.
-
All authors declare that they have no competing interests.
-
The online version contains supplemental information available at https://doi.org/10.65390/fdiv.2026.136002. Supplementary File 1 Supplementary materials to this study.